A Brief History
For centuries, cereals have been recognized as fundamental foods throughout the world. In particular, bread, being one of them, has always been considered an essential food group. The centrality of bread is probably best noted in the famous proverb that says 'bread and water', originating from the minimum food that had to be ensured in prisons. Wheat has undoubtedly been a significant food as well as a pivotal part of the Mediterranean diet.
However, as widespread as it may be, wheat cannot be tolerated by everyone. This inevitably leads to the discussion of gluten that stems from the mention of wheat sensitivity, but let's first define what gluten actually is before we get into the genetics and the evolution of things.
Gluten is the umbrella term for proteins that are found in wheat (wheatberries, durum, graham, emmer, semolina, spelt, farina, farro, KAMUT Khorasan wheat and einkorn), rye, barley and triticale (a cross between wheat and rye). Gluten ensures that foods maintain their shape by acting as a kind of glue that holds food together. Gluten is widespread and can be found in many types of foods.
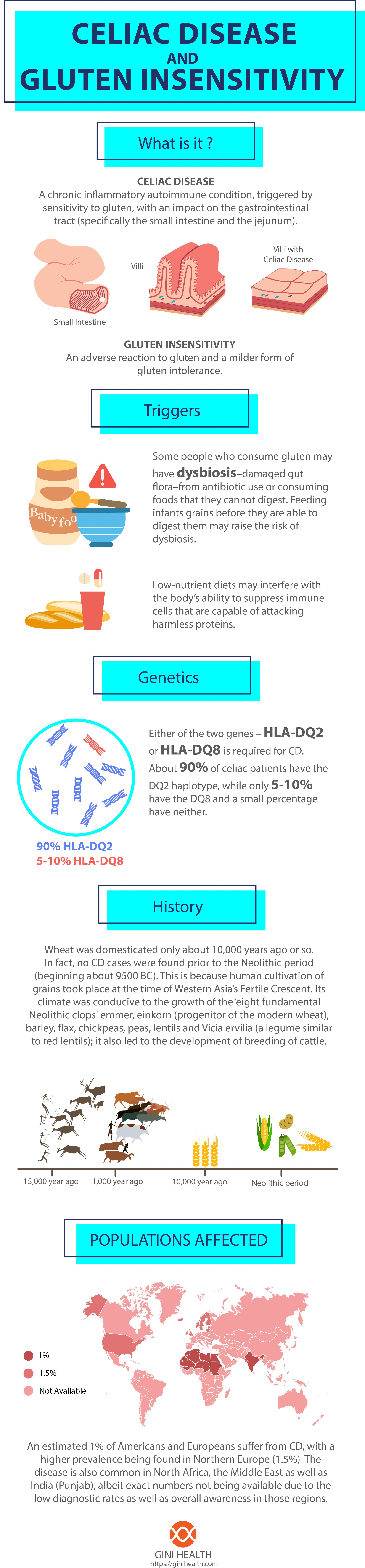
Probably the best known disease that is related to gluten consumption is celiac disease (CD), a chronic inflammatory condition with an impact on the gastrointestinal tract (specifically the small intestine and the jejunum) [1]. In particular, the absorbent apparatus within the small intestine is affected, whereby it atrophies, and this inadvertently leads to a deterioration of nutrient absorption [1].
The disease is well defined and characterized - the first case of it was reported in 250 AD, marked by symptoms of diarrhea, weight loss as well as general decline in children and young adults. For the purposes of this post, the main focus will be a milder form of CD, namely gluten or wheat insensitivity.
Celiac Disease and Evolution
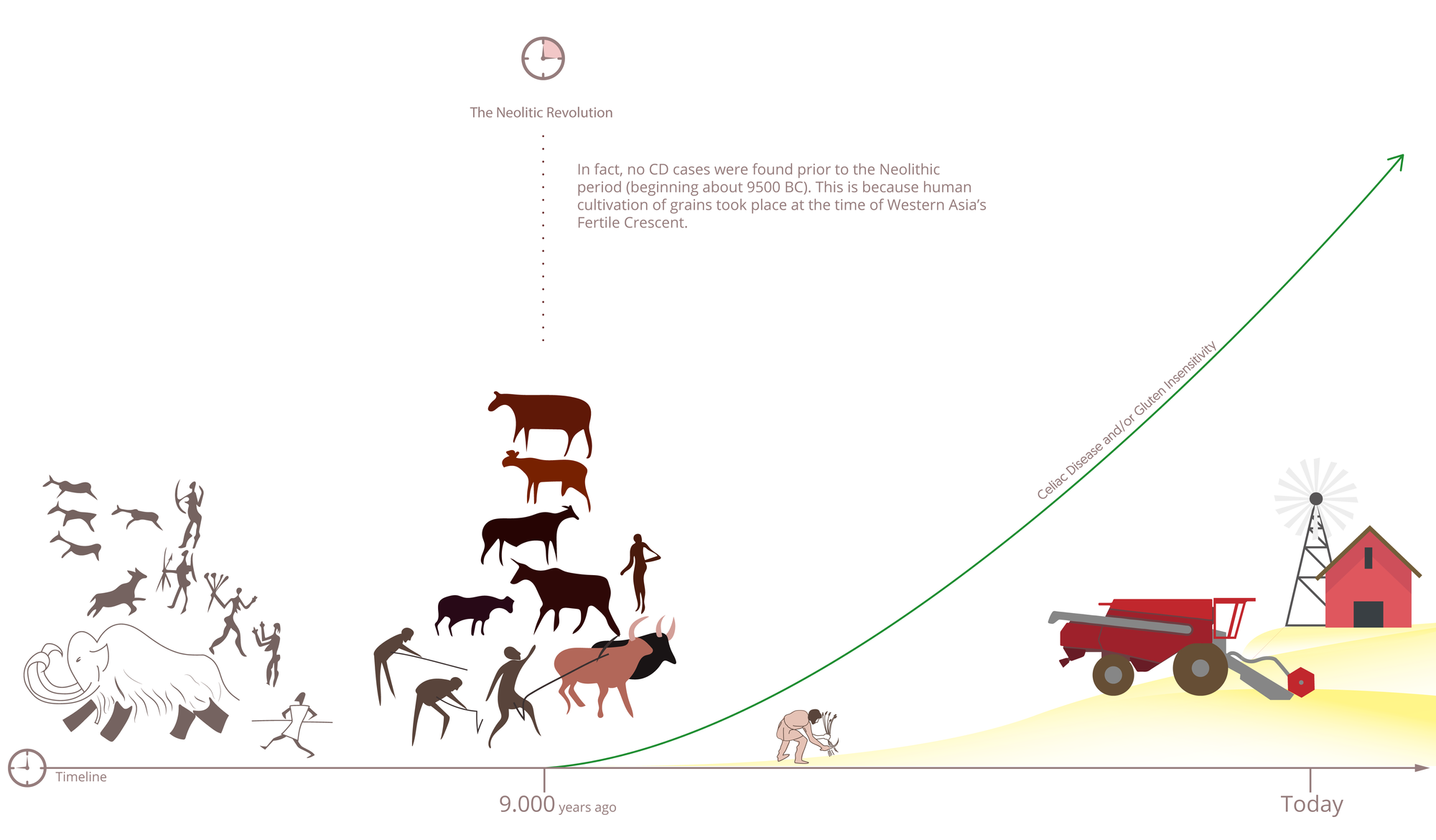
The most important events of human nutrition weigh back about 10,000 years ago, a time that was marked by domestication of animals such as dogs and horses. This was also a time that included breeding of sheep and cattle. Another nutritional milestone that took place around that time was the cultivation of seed also took off, and it brought with it the very first agriculture. In fact, it is speculated that the first agricultural civilizations were developed in Egypt as well as Mesopotamia. Fast forward a few thousand years, and one can see the rising of agriculture independently in many areas of the world.
While gluten has been a large component of our lives and diets today, it has not been the case for very long. Wheat was domesticated only about 10,000 years ago or so.
In fact, no Celiac Disease cases were found prior to the Neolithic period (beginning about 9500 BC). This is because human cultivation of grains took place at the time of Western Asia's Fertile Crescent [2].
Its climate was conducive to the growth of the 'eight fundamental Neolithic clops' emmer, einkorn (progenitor of the modern wheat), barley, flax, chickpeas, peas, lentils and Vicia ervilia (a legume similar to red lentils); it also led to the development of breeding of cattle.
Human knowledge and domains were expanded to Africa, Europe and the East, spanning to Australia and also perhaps South America. Agriculture was much slower in the remaining regions of the world - China was a few thousand years behind, where millet was grown in the north about 9000 years ago and rice cultivation began in the south at the same time. Central and South American agriculture beginnings date back to 8000 years ago with the cultivation of corn, squash and beans as well as growing potatoes, tomatoes, cacao and cassava.
Agriculture and livestock arrived in North America about 8000 - 9000 years ago and then it reached Africa.
There is also the famous "case of Cosa" that is a term referring to a skeleton dating back to the first century AD that was found at the archeological site of Cosa, southwest of Tuscany, Italy in 2008. The skeleton was that of a woman who is presumed to have been 18-20 years old - it showed signs of celiac disease damage (failure to thrive as well as malnutrition) in addition to the presence of human leukocyte antigen (HLA)-DQ2.5 (more about genetic markers of celiac disease in a bit) [3].
Celiac Disease - What is it?
Necessary conditions for Celiac Disease devlopment
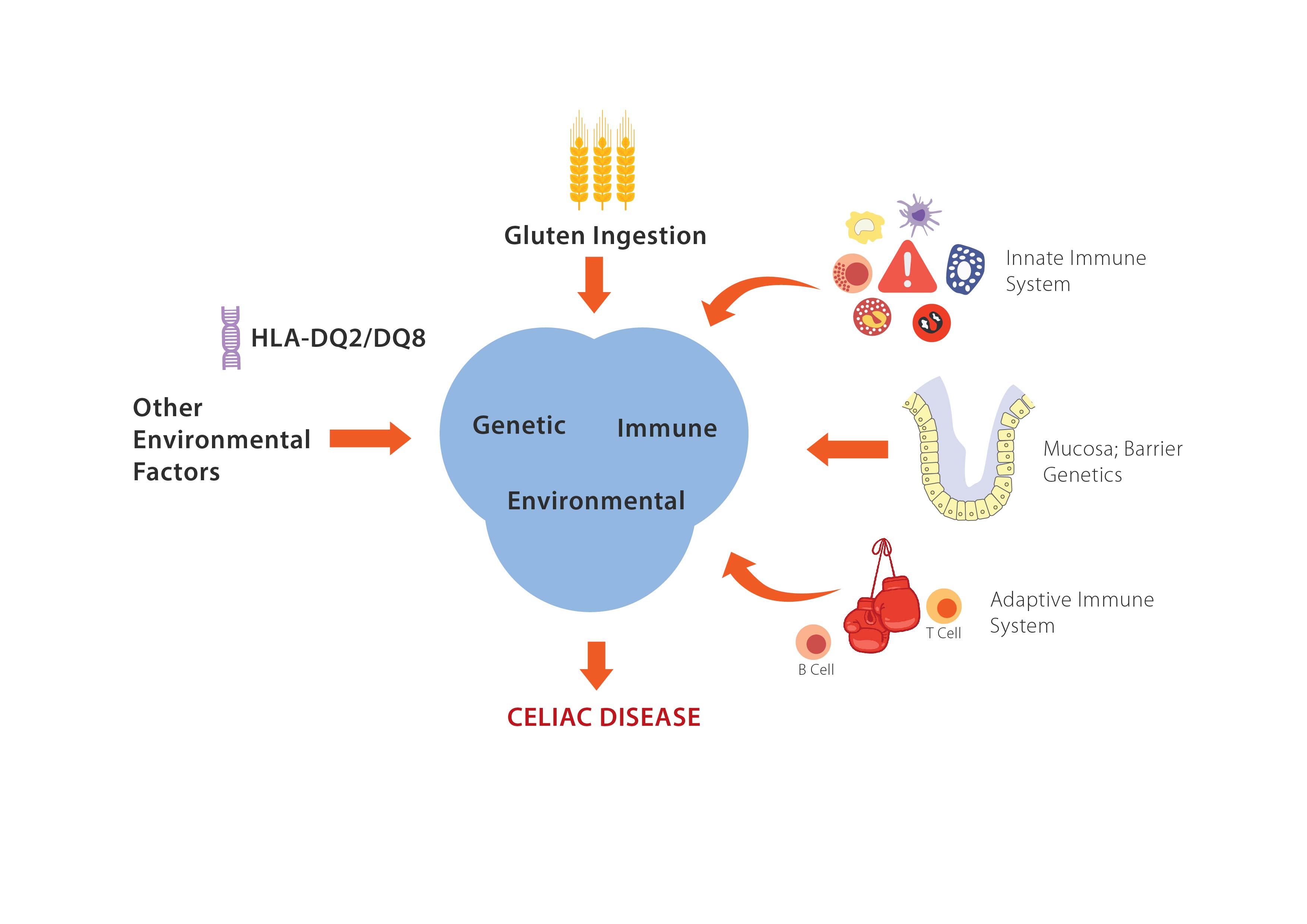
CD or gluten-sensitive enteropathy (GSE) is life-long intolerance to specific storage proteins in wheat (gliadin), rye (secalin) and barley (hordein), which are collectively called 'gluten'. It is a genetically based autoimmune disease in which gluten triggers the progressive damage to the mucosal villi of the small intestine [4]. It is characterized by malabsorption of nutrients that are absorbed in the proximal small bowel such as iron, folate and calcium. Disease progression along the intestine is marked by malabsorption of carbohydrate, fat and the fat-soluble vitamins A, D, E and K, and other micronutrients. Secondary lactose intolerance resulting from decreased lactase production by the damaged villi is also common [5] While CD symptoms can develop early in life following the introduction of gluten-containing cereals, it is very common for the disease to manifest itself during adulthood [4].
Powerful Gene
About 95% of celiac patients have the DQ2 haplotype, while only 5% have the DQ8 and a small percentage (about 2%) that have neither, identifiable by DQA10501/DQB10201 or DQA10501/DQB10202 and DQB1*0302 alleles. As with all genetic markers, the presence of the DQ2 and DQ8 is an important factor indicating susceptibility to CD, yet it cannot be considered the one and only indicating sign [6].
Only genetically predisposed individuals have the risk of suffering from celiac disease - meaning that if you have inherited the genetic mutations for it, you are more likely to have it.
Either of the two genes - HLA-DQ2 or HLA-DQ8 is required for CD. At the same time, having them also raises an individual's likeliness of suffering from non-celiac gluten sensitivity [7].
The function of the two HLA genes is pretty elegant - they essentially determine whether or not your body detects gluten in your system. However, they do not decide if your body will recognize it as harmless or as a foreign invader. In order for that to take place, the immune system must basically be fooled into recognizing pseudo-microbial-infection patterns as real ones.
Environmentally Speaking About Celiac Disease
So what must hold true in order for the body to recognize gluten as a foreign (and unwelcome) invader? There is not one clear answer yet, but some possible suggestions are as follows [7]:
- Some people may possess as-yet unidentified genes that cause their immune system to think an undigested fragment of the gluten protein looks like a microbial invader.
- Some people who consume gluten may have dysbiosis-damaged gut flora-from antibiotic use or consuming foods that they cannot digest. Feeding infants grains before they are able to digest them may raise the risk of dysbiosis. In this scenario, the immune system may see the products of microbial invasion from the dysbiosis and the undigested gluten fragment at the same time and be tricked into thinking that the gluten fragment is the microbial invader.(See below for an elaboration of environmental factors contributing to CD.)
- Low-nutrient diets may interfere with the body's ability to suppress immune cells that are capable of attacking harmless proteins. For example, one of the chemicals the body uses to suppress these immune cells is TGF-β, which is upregulated by vitamin A. A diet deficient in vitamin A, then, might undermine the body's ability to keep its immune system from attacking harmless proteins like gluten. (See below for an elaboration of environmental factors contributing to CD.)
CD is triggered by virtue of ingesting gluten. This in and of itself is an environmental trigger in those with a genetic predisposition to the disease. Some of the environmental factors that are considered as contributors to CD include quantity and quality of gluten that is ingested, the kind and length of wheat dough fermentation, early infant feeding, intestinal microorganisms and how they evolve over time, intestinal infections and overall stressors [8].
In terms of infant feeding, in particular, studies have shown that delaying the introduction of gluten in a child's life may have two positive effects, namely developmental delay of CD as well as a reduced frequency in the HLA-high-risk group [9]. When it comes to breastfeeding, no protective effects have been found in children that were breastfed [9].
And while the aforementioned still remain to be fully confirmed as environmental factors that contribute to the increased chances of CD development, the concept of gut dysbiosis as a potential contributing factor has been studied more extensively. Increasing evidence points to the role of gut microbiota in the regulation of intestinal immune response as well as maintenance of intestinal homeostasis. Several studies have confirmed altered gut microbiota composition or function in persons with CD versus healthy individuals [10, 11, 12, 13, 14, 15, 16].
Environmental factors that influence a disbalance in the composition of microbiota include the type of milk that is ingested (breast milk vs. formula), type of delivery, exposure to antibiotics as well as other factors that profoundly affect intestinal health [17]. However, further studies are warranted that confirm whether it is indeed intestinal dysbiosis that contributes to CD by itself or a secondary consequence. [18].
Ethnicity and Celiac Disease
Even though previously thought to be an exclusively European condition, it is now known that CD is distributed worldwide [4].
An estimated 1% of Americans and Europeans suffer from CD, with a higher prevalence being found in Northern Europe (1.5%) [19, 20]. The disease is also common in North Africa, the Middle East as well as India (Punjab), albeit exact numbers not being available due to the low diagnostic rates as well as overall awareness in those regions.
A fairly recent study conducted on individuals living in the US (studied ethnicities include North Indian, South Indian, East Asian, Hispanic, Middle Eastern, Jewish and other Americans) has found that the condition was most common among those whose ancestors came from India's Punjab region, which is contrary to the previous assumption those with European ancestry are mostly affected [21]. Along with that finding, the researchers also reported that CD is much less common among Americans from South Indian, East Asian and Hispanic ancestry [21]. Furthermore, those with Jewish and Middle Eastern ethnicities had similar CD rates as other Americans [21].
Not Quite Celiac Disease but Still an Issue - Gluten Sensitivity
To be clear, gluten intolerance is not a food allergy, but rather a physical condition of the gut. Basically what that means is that undigested gluten proteins remain in your gut and are treated like foreign objects, irritating their surroundings (the gut) and flattening the microvilli along the wall of the small intestine [22].
With fewer microvilli available to absorb the nutrients from the food, those who are affected experience symptoms of malabsorption. These include chronic fatigue, neurological disorders, nutrient deficiencies, anemia, nausea, skin rashes, depression, and more [22].
Specifically, non-celiac gluten sensitivity is defined as sensitivity to food items that contain gluten in individuals in whom CD and wheat allergy have been ruled out [23]. A diagnosis of non-celiac gluten sensitivity is very specific - it involves the above mentioned symptoms along with a negative serology, negative immune-allergy test, normal duodenal biopsy and improvement of symptoms during a gluten-free diet [24].
There is a proposed final step in the identification or confirmation of non-celiac gluten sensitivity, which is basically a returning of all of the symptoms upon the introduction of wheat in the diet [25]. However, depending on the severity of the condition - ranging from a gluten sensitivity to CD - it may be possible to eventually consume grains again. These, however, ought to be properly prepared (sourdough that has fermented for up to a month or sprouted grains, as examples).
Even though CD is still relatively rare compared to the frequency of other diseases, it is advancing rapidly. Many (read millions) Americans with CD and gluten intolerance who must ensure that they are not consuming any gluten. This means that they are spending A LOT of time reading labels in supermarket aisles.
Is Gluten Intolerance in Your Genes?
There's an interaction between 6 SNPs to predict the risk for Celiac Disease. I have built a formula to assess that. You will need to upload your data at https://www.healthbit.co/23andme-users/ or email me at [email protected]. I will give you detailed nutrition report too. It can take few days before I send your report as I have many requests coming in. But it's totally free for you.
If you are intereted in knowing more about genetics an nutrition you can ask for an invitation at https://ginihealth.com/.
You can always email me at [email protected] to talk more about the mission to make nutrition personalized for everybody to increase the healthspan along with the lifespan.
References:
-
Gasbarrini, G.B. and Mangiola, F. (2014). Wheat-related disorders: A broad spectrum of 'evolving' diseases. United European gastroenterology journal, 2(4), pp.254-262.
-
Mocan, O. and DUMITRAŞCU, D.L. (2016). The broad spectrum of celiac disease and gluten sensitive enteropathy. Clujul Medical, 89(3), p.335.
-
Gasbarrini, G., Miele, L., Corazza, G.R. and Gasbarrini, A. (2010). When was celiac disease born?: the Italian case from the archeologic site of Cosa. Journal of clinical gastroenterology, 44(7), pp.502-503.
-
Fasano, A. and Catassi, C. (2001). Current approaches to diagnosis and treatment of celiac disease: an evolving spectrum. Gastroenterology, 120(3), pp.636-651.
-
Ojetti, V., Nucera, G., Migneco, A., Gabrielli, M., Lauritano, C., Danese, S., Zocco, M.A., Nista, E.C., Cammarota, G., De Lorenzo, A. and Gasbarrini, G. (2005). High prevalence of celiac disease in patients with lactose intolerance. Digestion, 71(2), pp.106-110.
-
Megiorni, F. and Pizzuti, A. (2012). HLA-DQA1 and HLA-DQB1 in Celiac disease predisposition: practical implications of the HLA molecular typing. Journal of biomedical science, 19(1), p.88. https://jbiomedsci.biomedcentral.com/articles/10.1186/1423-0127-19-88
-
What No One Is Saying About Zonulin - Is Celiac About More Than Genes and Gluten? (2011, April 6). Retrieved from https://chrismasterjohnphd.com/2011/04/06/what-no-one-is-saying-about-zonulin-is/
-
Lionetti, E. and Catassi, C. (2011). New clues in celiac disease epidemiology, pathogenesis, clinical manifestations, and treatment. International reviews of immunology, 30(4), pp.219-231.
-
Lionetti, E., Castellaneta, S., Francavilla, R., Pulvirenti, A., Tonutti, E., Amarri, S., Barbato, M., Barbera, C., Barera, G., Bellantoni, A. and Castellano, E. (2014). Introduction of gluten, HLA status, and the risk of celiac disease in children. New England Journal of Medicine, 371(14), pp.1295-1303.
-
Cenit, M.C., Olivares, M., Codoñer-Franch, P. and Sanz, Y. (2015). Intestinal microbiota and celiac disease: cause, consequence or co-evolution?. Nutrients, 7(8), pp.6900-6923.
-
Verdu, E.F., Galipeau, H.J. and Jabri, B. (2015). Novel players in coeliac disease pathogenesis: role of the gut microbiota. Nature Reviews Gastroenterology and Hepatology, 12(9), p.497.
-
Cheng, J., Kalliomäki, M., Heilig, H.G., Palva, A., Lähteenoja, H., de Vos, W.M., Salojärvi, J. and Satokari, R. (2013). Duodenal microbiota composition and mucosal homeostasis in pediatric celiac disease. BMC gastroenterology, 13(1), p.113.
-
Sánchez, E., Donat, E., Ribes-Koninckx, C., Fernández-Murga, M.L. and Sanz, Y. (2013). Duodenal-mucosal bacteria associated with celiac disease in children. Applied and environmental microbiology, 79(18), pp.5472-5479.
-
Sellitto, M., Bai, G., Serena, G., Fricke, W.F., Sturgeon, C., Gajer, P., White, J.R., Koenig, S.S., Sakamoto, J., Boothe, D. and Gicquelais, R. (2012). Proof of concept of microbiome-metabolome analysis and delayed gluten exposure on celiac disease autoimmunity in genetically at-risk infants. PloS one, 7(3), p.e33387.
-
Di Cagno, R., De Angelis, M., De Pasquale, I., Ndagijimana, M., Vernocchi, P., Ricciuti, P., Gagliardi, F., Laghi, L., Crecchio, C., Guerzoni, M.E. and Gobbetti, M. (2011). Duodenal and faecal microbiota of celiac children: molecular, phenotype and metabolome characterization. BMC microbiology, 11(1), p.219.
-
Collado, M.C., Donat, E., Ribes-Koninckx, C., Calabuig, M. and Sanz, Y. (2008). Imbalances in faecal and duodenal Bifidobacterium species composition in active and non-active coeliac disease. BMC microbiology, 8(1), p.232.
-
Mårild, K., Ye, W., Lebwohl, B., Green, P.H., Blaser, M.J., Card, T. and Ludvigsson, J.F. (2013). Antibiotic exposure and the development of coeliac disease: a nationwide case-control study. BMC gastroenterology, 13(1), p.109.
-
Lionetti, E. and Catassi, C. (2015). The Role of Environmental Factors in the Development of Celiac Disease: What Is New?. Diseases, 3(4), pp.282-293.
-
Mahadov, S. and Green, P.H. (2011). Celiac disease: a challenge for all physicians. Gastroenterology & hepatology, 7(8), p.554.
-
Gujral, N., Freeman, H.J. and Thomson, A.B. (2012). Celiac disease: prevalence, diagnosis, pathogenesis and treatment. World journal of gastroenterology: WJG, 18(42), p.6036.
-
People Whose Ancestors Came From This Region May Be More Likely to Have Celiac Disease. (2016, June 1). Retrieved from http://www.health.com/nutrition/researchers-uncover-surprises-about-celiac-disease)
-
The Rise of Gluten Intolerance. (n.d.) Retrieved from https://www.foodrenegade.com/the-rise-of-gluten-intolerance/
-
Ludvigsson, J.F., Leffler, D.A., Bai, J.C., Biagi, F., Fasano, A., Green, P.H., Hadjivassiliou, M., Kaukinen, K., Kelly, C.P., Leonard, J.N. and Lundin, K.E.A. (2013). The Oslo definitions for coeliac disease and related terms. Gut, 62(1), pp.43-52.
-
Siniscalchi, M., Iovino, P., Tortora, R., Forestiero, S., Somma, A., Capuano, L., Franzese, M.D., Sabbatini, F. and Ciacci, C. (2005). Fatigue in adult coeliac disease. Alimentary pharmacology & therapeutics, 22(5), pp.489-494.
-
Sanders, D.S., Evans, K.E. and Hadjivassiliou, M. (2010). Test for coeliac disease first. BMJ, 341:c5161.